All you sci-fi enthusiasts out there will be familiar with the common trope of the mirror self; a seemingly identical version of a character except that they exist in a mirrored universe. These mirror characters, in addition to being a physical mirror, are often a psychological mirror as well; evil where their original versions are good. The idea that your mirror self could have ‘opposite’ personality traits to your own, although a fun construct, runs counter to our intuition about how consequential something like a geometrical inversion should be.
Why should it matter that a person’s mole is on the other side of their face, or their hair parts in the other direction? However, there are many examples of natural objects which have mirror symmetric versions and just like in the fiction trope, this seemingly arbitrary distinction can have some surprising consequences.
What is mirror symmetry?
Objects which exhibit mirror symmetry are referred to as ‘chiral’. This term comes from the Greek word for hand, a clear origin, since your hands are the perfect example of two mirror symmetric objects. If you take a look at your hands, you’ll see that no matter how you turn them, you cannot lay them so they are superimposed, despite being otherwise identical. Instead, if you hold one up to a mirror, this image, which has been reflected in the plane of the mirror, will be identically orientated to the other hand.
To discriminate between the two versions we use the terms left- and right-handed. A simple way to identify which version is which is to look at the palms of your hands, if the thumb is on the left of the index finger then this is the left hand and vice versa. Scientists have similar rules which they use to identify left- and right-handed versions of the objects they’re studying.
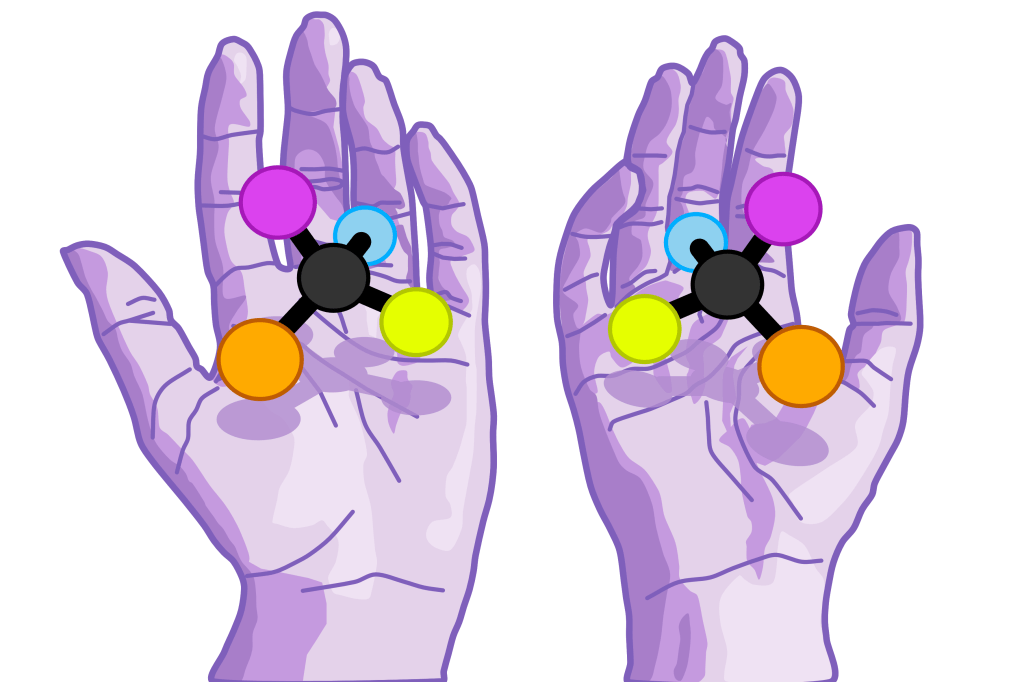
Chirality can be seen at every level of nature, from the most fundamental aspects of physics, to macroscopic examples such as the direction that a plant’s leaves spiral around its stem. Chirality is a surprisingly important aspect to DNA, molecules that we can taste and smell, and the way that subatomic particles behave. It has also had a tragic role in the development of certain medicines. Let’s take a look at each of these examples in more detail.
Chiral spiral – the handedness of DNA
DNA is a fundamental building block of life. We all have a pretty good idea of what DNA is, and a large part of that is thanks to the determination of its shape in the mid-1900s. Now, most people would recognise the unique helical structure of DNA.
Have you ever stopped to ponder the direction of this immediately recognisable spiral though? Perhaps the handedness of the twist has never occurred to you (as pointed out by science illustration cooperative SquareCell, DNA is also often misrepresented, even in scientific media), but in fact the majority of DNA spirals in a clockwise direction, which is equivalent to being right-handed.
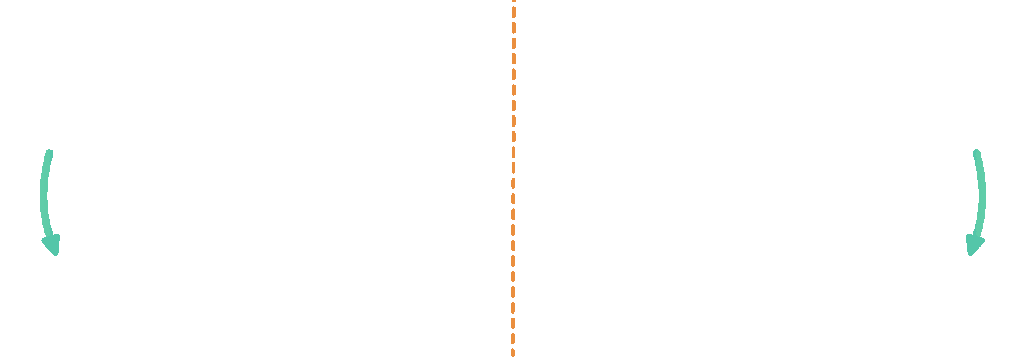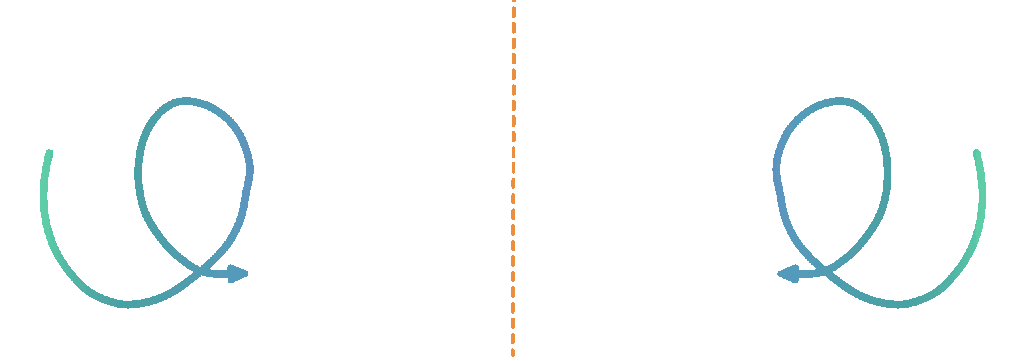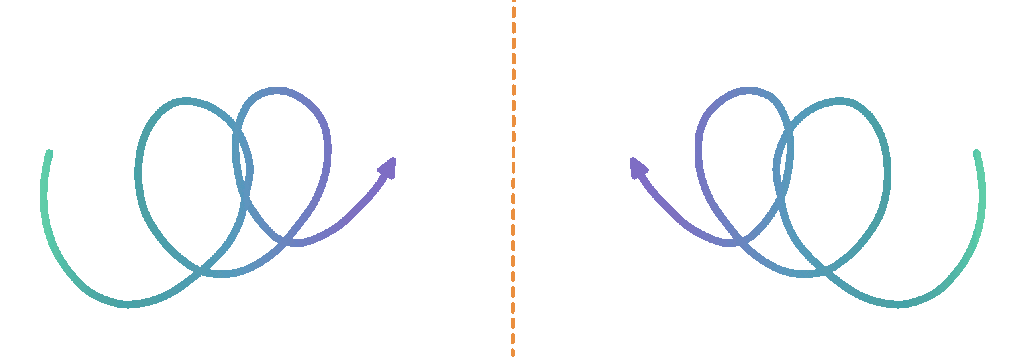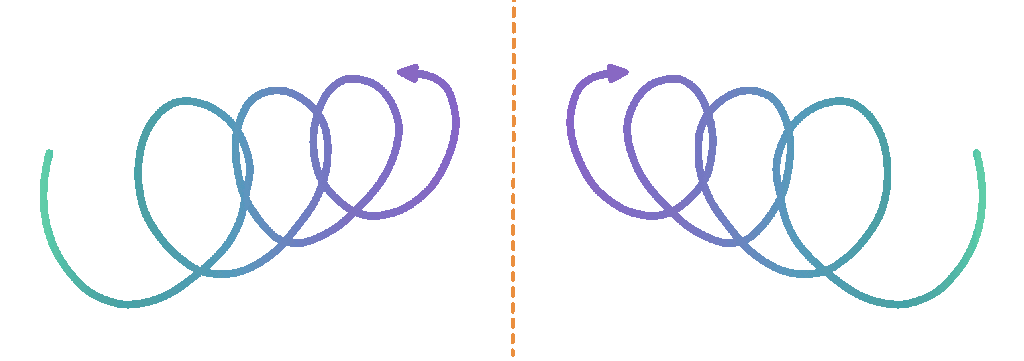
Scientists have been trying to understand the origin of this asymmetry since it was first identified. It seems that somewhere in history some mechanism created an imbalance in the relative amounts of each helical version. Most likely, this happened extremely early because the self-replicating nature of DNA means that its chirality is maintained; each new DNA molecule twists in the same direction as its parent.
Essentially, there are two potential origins of the imbalance in DNA chiralities: either the preference occurred by chance as a small difference between the two options and was magnified over time, or there exists some process that DNA participates in which always favours one handedness over the other.
If chance was the culprit, then the explanation could be as simple as basic probability; each DNA strand in the initial mixture would have had a fifty-fifty chance of being left or right-handed, just like a coin toss. If you only make a few pieces of DNA, or toss a coin a few times, you are likely to end up with a small excess of one result over the other. Alternatively, there may have been some random outside influence which created a small excess.
In either case, once a small imbalance had formed, there needed to be some reason that it was magnified, such that eventually only one handedness prevailed. There are some proposed mechanisms which could explain how this happened. However, a very interesting idea, which does not rely on a single, small excess at the beginning of life, has been under consideration for some time, waiting for a likely mechanism.
Scientists interested in the origins of chiral asymmetry in biology have long wondered if it is caused by a similar asymmetry in fundamental physics, to be discussed below. However, there hasn’t been a satisfying explanation which can link the two. In 2020, Dr Noemie Globus and Dr Roger D. Blandford at New York University published a paper suggesting a direct link between the asymmetry in the weak charge, to be discussed below, and the single spiral direction found in DNA. This is a pleasing result from an aesthetic standpoint, because it links fundamental physics to the biological world, but it has not yet been experimentally tested.
The link between the asymmetry in DNA helices and the weak charge comes from the fact that many of the mutations that allow organisms to evolve are created when a cosmic ray collides with a DNA strand. Collisions cause damage that can lead to a mutation and this mutation may be propagated to future generations if it is advantageous to the survival of the organism – the foundation of Darwin’s theory of evolution.
If cosmic rays exist in one chirality only and this chirality is more likely to damage DNA of one handedness, then it follows that this type of DNA will become dominant because the organisms it belongs to will be able to adapt more efficiently to their environments. Globus and Blandfords’ paper uses theoretical modelling to show that this theory has merit.
Philosophy of science academics will tell you though, that most of the time a good scientific theory is one which you can use to make testable predictions. The cosmic ray theory has that benefit; for example if you took two identical colonies of bacteria and subjected one to the same kind of radiation as is found in cosmic rays, you would expect to see a higher mutation rate in that colony compared to the other. I look forward to seeing the results of this experiment one day!
When mirrors smell different
Another strange example of mirror-image molecules behaving differently, occurs when they are involved in taste and smell. In this case, the peculiarity is not to do with the molecules themselves but rather how they interact with our bodies.
Taste and smell are both chemo senses meaning that what we are sensing when we smell or taste something are small quantities of molecules that are important for our health; either because they are found in our food, tell us about other humans, or they function as a warning system.
The way our bodies sense chemicals is nifty. Inside our nose, olfactory neurons in the skin lining our airways, extend their dendrites all the way down to sample the air rushing by. On their ends are little protrusions called cilia which contain proteins that bind only to specific molecules. A binding event triggers the neuron to send a signal up through our nose, into our olfactory bulb and then to our brain where we experience the odour. Interestingly, a lot of our sense of taste also occurs in our nose because out taste buds can only discriminate between saltiness, bitterness, sweetness, sourness, and savouriness. When we eat, molecules from our food fly up the back of our throat into our nasal cavity, where they are detected in the same way as smells.
In some cases, the chemicals we sense are chiral. How we interpret the two chiralities can vary a lot; for some molecules, the two versions are exactly the same, for others they only differ in their intensity, and in others still they are interpreted as completely different. An interesting example of a molecule with two totally different tasting chiralities is the chemical carvone. The two chiral versions of carvone, referred to using the chemical notation of D and L, are present in different plants. D-carvone is found in caraway and dill, while L-carvone is found in spearmint. Pretty different tastes! Another example are the amino acids, D-amino acids usually taste sweet, whereas some L-amino acids are bitter.
Unfortunately, it is not yet fully understood what happens when chemicals interact with our chemosensory receptor proteins. Scientists suspect there is a complex combination of two processes which lead to this variable response across different chiral molecules.
The first process is the lock and key interaction which you might have learned about in school when you studied enzymes. Essentially, the enzyme or protein has a binding site that is uniquely shaped so that only one thing can fit into the space. However, if this were the sole mechanism in chemosensation then chiral molecules would either always be interpreted as different or we would only be able to sense one chirality.
Instead of being dependent on shape, the interaction could rely on the transfer of a specific amount of energy. In this case the chirality of the molecule should have no effect and both chiralities would always taste or smell the same.
Given that neither of these explanations can account for the variable way that oppositely handed molecules can smell or taste, there must be more complexity to this interaction than we first assumed. For example, a paper published in 2009 suggests that the various ways chiral molecules can be interpreted has to do with their rigidity. Counterintuitively, they found that molecules which are rigid, that is their shape can’t be ‘bent’, are those whose mirror images smell the same. On the other hand, flexible molecules seem to be those whose mirror images smell different. The interpretation for why this could work is complex, however, it’s an interesting idea which deserves further investigation.
The sad story of thalidomide
Differences in how mirror symmetric molecules smell provide a fun example of the unique ways our body can respond to chirality. However, not all examples are innocuous; when pharmaceutical drugs are involved, it can be disastrous if the two mirror image versions of that drug to behave differently in the body.
If you have had a baby or know someone who has, then you are aware that it can be an unpleasant experience, particularly at the beginning of the pregnancy. In the first trimester pregnant people often feel very nauseated and may even vomit. Often called morning sickness, this unpleasant side-effect of pregnancy is due to the change in hormone levels that occurs when conception is achieved.
Naturally, pregnant people would prefer to avoid feeling constantly sick for three months, so anti-nausea medication has a large market. This demand was met in the late 1950s when the drug thalidomide was sold over the counter. Initially developed as an antihistamine, thalidomide turned out to be more effective at inducing drowsiness and was therefore marketed as a sleep-aid, anti-anxiety medication, and for morning sickness – you can’t feel sick if you’re asleep?
Only a few years after it was first marketed, it became obvious that thalidomide was very dangerous for developing babies. If a mother took thalidomide herself, or the baby was conceived with a father who was taking it, then the baby could be born with birth defects such as short or even absent limbs, or abnormalities to the ears, eyes, heart or gut. Even a single dose could be enough to have these impacts.
Just like other chiral molecules the two types of thalidomide are referred to with letter prefixes; S-thalidomide is responsible for the detrimental effects during pregnancy, whereas R-thalidomide makes you sleepy.
These observed effects are somewhat paradoxical however, as each chirality, when taken in isolation, can actually be converted to its mirror version when absorbed into the body. So how can experiments have shown that taking a pure version of R-thalidomide does not cause birth defects? Recent work identified a possible explanation by replicating the conditions inside the body. In this specific environment, they saw that an initial excess of either S- or R-thalidomide will be amplified over time. This is because although some of the molecules will be converted to their mirror versions, these inverted molecules would form a pair with one of their mirrors. Pairs were no longer soluble and would form a solid that fell to the bottom of the experimental vessel making them unable to participate in further interactions and effectively inert. If this process is borne out in a similar way inside the body, then it explains why ingesting R-thalidomide does not lead to birth defects.
The story of thalidomide is not entirely tragic. Although it was rapidly removed from sale as an anti-nausea medication in 1961, thalidomide also has some genuine uses for treating cancer and other diseases. So, it is still occasionally prescribed under strict guidance.
Mirrored Particles?
The examples given so far have all been relatively macroscopic. That is, they have been about molecular chirality, and although molecules are very small by human standards, they are still large from a physicist’s point of view! However, given that a mirror reflection is one of the most fundamental symmetries found in geometry, it is not surprising that we can also find mirror symmetry effects in even the tiniest parts of nature.
At the smallest of scales, the universe can be described by the standard model of particle physics. Here, when physicists consider mirror images they don’t use the word ‘chirality’, instead they have another fancy word: parity. The term parity is usually applied to an object in the same way that right- or left-handed is, i.e a particle has a particular parity. A particle’s parity is determined by the relative orientation of the characteristics of ‘momentum’, and ‘spin’, in the same way that hands are identified by which side the thumb is compared to the index finger.
Just like with the interesting differences seen in chiral molecules, physicists were surprised to learn that their initial assumptions about fundamental particles of opposite parity were wrong. In the 1950s, theoretical physicists Tsung-Dao Lee and Chen-Ning Yang realised that while parity had been shown to be inconsequential for certain types of particle physics, it had not been experimentally tested in all cases. This led to an experiment headed by Professor Chien Shiung Wu, which showed that the parity of subatomic particles taking part in certain interactions was actually very important.
To understand specifically what Wu and her colleagues had found, we need to take a quick detour to discuss the types of matter in our universe. Everything that we would consider the matter that makes up the universe is actually just one side of a coin; there are in fact equivalent versions of each of ‘our’ subatomic particles, known as antiparticles. These antiparticles have the same mass but differ in certain aspects, such as their electric charge. Electric charge is the only type of charge that we have any everyday experience with because it’s what we harness to make electricity, but there are other types of charge as well.
In Wu’s experiment, the charge under consideration was the weak charge, which is important for the weak interaction between matter particles. The weak interaction is another fundamental force of nature like the electromagnetic force or gravity, but it is very weak, as you might guess from the name. Wu’s experiment led to the understanding that a weak charge can only be assigned to left-handed particles and right-handed antiparticles, which means the other versions cannot take part in weak interactions.
This is a confusing concept but the essential take-away is that when weak interactions occur, only matter particles/antiparticles with one parity (or handedness) can participate. This is indeed a very strange result; imagine if the laws of physics prevented you from ever throwing a ball with your right-hand!
Wu’s experiment showed that at the most fundamental level of nature, just like in the sci-fi trope, the two mirrored versions of an object can actually behave differently. You might ask then, what other assumptions have we discovered were incorrect? And what other consequences has this had, aside from perhaps the handedness of DNA?
Same but different
Despite being a seemingly innocuous and inconsequential effect of a geometrical symmetry, there are many examples where the mirrored version does in fact behave quite differently. Perhaps there is merit to the sci-fi trope after all.